STZ-induced diabetic mouse model, a recent study showed that a novel DPP-IV inhibitor can ameliorate diabetes by increasing b-cell TWS119 replication and neogenesis. Another study also showed that a novel, potent, specific and substrate-selective DPP-IV inhibitor can improve glycemic control and b-cell damage. The restorative effects of those DPP-IV inhibitors following STZ injury on pancreatic b-parameters were overall consistent with our DPP-IV inhibitor, sitagliptin treatment. Furthermore, we found that combining a GPR119 agonist with a DPP-IV inhibitor is significantly better than either alone. Because GPR119 is expressed  on pancreatic b-cells as well as intestinal L cells, it is possible that PSN632408 could have improved blood glucose levels and increased pancreatic b-cell mass by a direct action on b-cells and/or by stimulating GLP-1 production by intestinal L cells. These alternatives might be answered by using GLP-1 receptor knockout mice. Multiple factors may contribute to the effects on pancreatic bcell mass including b-cell regeneration, hypertrophy and apoptosis. To assess b-cell regeneration in these mice, we continuously labeled mice with BrdU to track replicating cells and measured the proliferation rates in terms of percentage of insulin and BrdU co-positive cells in total islet bcells. We found that PSN632408 and sitagliptin combination treatment significantly increased the numbers of replicating b-cells compared with vehicle or PSN632408 treatment alone. It has been suggested that BrdU incorporation is associated with a DNA damage response, not replication, in human pancreatic b-cells. Therefore, we did not solely rely upon BrdU incorporation as evidence of b-cell replication. We used Ki67, a cellular marker for replication, to AB1010 further determine b-cell replication. Ki67 is strictly associated with cell replication and is expressed during all phases of the cell cycle tracking active dividing cells. Although Ki67 staining would have identified the cells undergoing cell division during the last fraction of the treatment period, our results corroborated a similar trend observed with insulin and BrdU staining. Using insulin and Ki67 staining as reliable evidence of bcell replication, we found that treatment with PSN632408 alone or sitagliptin alone could stimulate b-cell replication; however, PSN632408 and sitagliptin combination was significantly better than either alone. Whether the replication of these b-cells was from self-renewal of mature b cells or was from specialized progenitors in islets needs to be further investigated. There is compelling evidence supporting b-cell neogenesis from precursors/stem cells in the ductal epithelium of the pancreas as a mechanism of b-cell regeneration in several diabetic models. Exendin-4, a GLP-1 analogue, has been shown to stimulate not only b-cell replication, but also b-cell neogenesis. In this study, we observed insulin positive cells located in the epithelial cell lining of pancreatic ducts. These insulin positive cells lining ducts were further confirmed to be exocrine duct cells, using CK-19, a ductal epithelial cell marker. Although, considerable animal to animal heterogeneity was observed across all treatment groups, mice treated with either PSN632408 alone or PSN632408 and sitagliptin combination showed significant increases in insulin/CK19 co-positive duct cells. We did not detect any glucagon positive cells in pancreatic ducts.
on pancreatic b-cells as well as intestinal L cells, it is possible that PSN632408 could have improved blood glucose levels and increased pancreatic b-cell mass by a direct action on b-cells and/or by stimulating GLP-1 production by intestinal L cells. These alternatives might be answered by using GLP-1 receptor knockout mice. Multiple factors may contribute to the effects on pancreatic bcell mass including b-cell regeneration, hypertrophy and apoptosis. To assess b-cell regeneration in these mice, we continuously labeled mice with BrdU to track replicating cells and measured the proliferation rates in terms of percentage of insulin and BrdU co-positive cells in total islet bcells. We found that PSN632408 and sitagliptin combination treatment significantly increased the numbers of replicating b-cells compared with vehicle or PSN632408 treatment alone. It has been suggested that BrdU incorporation is associated with a DNA damage response, not replication, in human pancreatic b-cells. Therefore, we did not solely rely upon BrdU incorporation as evidence of b-cell replication. We used Ki67, a cellular marker for replication, to AB1010 further determine b-cell replication. Ki67 is strictly associated with cell replication and is expressed during all phases of the cell cycle tracking active dividing cells. Although Ki67 staining would have identified the cells undergoing cell division during the last fraction of the treatment period, our results corroborated a similar trend observed with insulin and BrdU staining. Using insulin and Ki67 staining as reliable evidence of bcell replication, we found that treatment with PSN632408 alone or sitagliptin alone could stimulate b-cell replication; however, PSN632408 and sitagliptin combination was significantly better than either alone. Whether the replication of these b-cells was from self-renewal of mature b cells or was from specialized progenitors in islets needs to be further investigated. There is compelling evidence supporting b-cell neogenesis from precursors/stem cells in the ductal epithelium of the pancreas as a mechanism of b-cell regeneration in several diabetic models. Exendin-4, a GLP-1 analogue, has been shown to stimulate not only b-cell replication, but also b-cell neogenesis. In this study, we observed insulin positive cells located in the epithelial cell lining of pancreatic ducts. These insulin positive cells lining ducts were further confirmed to be exocrine duct cells, using CK-19, a ductal epithelial cell marker. Although, considerable animal to animal heterogeneity was observed across all treatment groups, mice treated with either PSN632408 alone or PSN632408 and sitagliptin combination showed significant increases in insulin/CK19 co-positive duct cells. We did not detect any glucagon positive cells in pancreatic ducts.
Month: August 2019
Cells are derived progenitors or from another source should be further determined using lineage-tracing experiments
Also, monitoring PDX-1 expression at different stages of the treatment period may answer whether or not these ductal cells are contributing to islet neogenesis. It is well known that b-cell replication strictly declines with age in mice and in humans. This phenomenon might be due to down regulation of key transcription factors and kinases implicated in b-cell mitosis. In our studies, the mice were,10 weeks old after diabetes induction and the mice were treated for 7 weeks. These mice were not aged mice, hence the replicative pool of cells would be considered abundant. Interestingly, the rate of b-cell replication in the pancreas of STZ-induced diabetic mice treated with PSN632408 was lower than the rate of b-cell replication in islet grafts in STZ-induced diabetic mice treated with PSN632408 in our earlier study. We do not know why PSN632408 could stimulate more b-cell replication in intact islet grafts. One possibility is that STZ demolished a lot of b-cells in the pancreas that have the capability to replicate. Another possible reason is that b-cells in intact islet grafts replicate more in a high glucose milieu, since almost all recipient mice had blood glucose levels.600 mg/dL before islet transplantation. Further studies are needed to determine whether b-cell replication is from self-renewal of mature b cells or from replication of specialized progenitors and whether different glucose Cycloheximide company levels affect b-cell replication in mice treated with PSN632408 and sitagliptin. In addition to b cells, we found a more than 2-fold increase in replication of a-cells when mice were treated with PSN632408 or sitagliptin alone, and more than a 5-fold increase when treated with combination therapys; however, a-cell mass was not measured. Alpha-cell replication and elevated Epoxomicin Proteasome inhibitor glucagon levels may aid in the formation of new b-cells, since pancreatic glucagon is required for b-cell formation and differentiation. Also, the composition of a-cells increases in pancreatic islets of diabetic human patients and of animal models. Interestingly, acells can be converted into b-cells under conditions of extreme damage to b-cells. Therefore, we cannot rule out the possibility that DPP-IV inhibitors or GPR119 agonists might play a role in a- tob-cell differentiation. There is some evidence for contribution of acinar cells in islet b-cell formation by transdifferentiation under specific conditions ; however, we did not find any increase in exocrine cell replication in any treatment group. To the best of our knowledge, this is the first 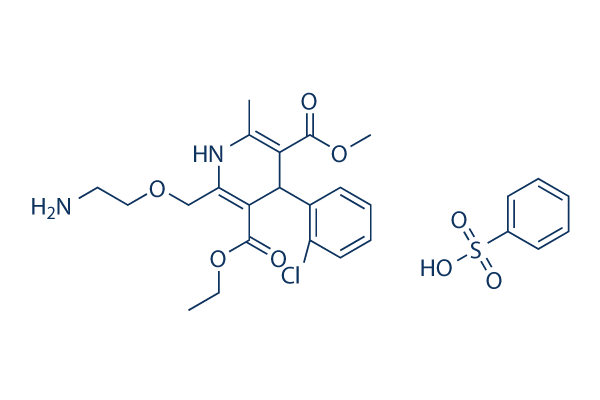 study demonstrating the effects of a GPR119 agonist along with a DPP-IV inhibitor on b-cell regeneration via both replication and neogenesis in a diabetic mouse model. Besides b-cell regeneration, other factors including prevention of b-cell apoptosis may have also contributed to the increase of b-cell mass. Pancreatic b-cell mass was remarkably improved by combination therapy, which offers a novel therapeutic strategy for treating diabetic patients with a low b-cell mass. As there are clear discrepancies between rodent and human b-cell regeneration capacity, our future studies are aimed at evaluating the potential of these drug combinations on human islet regeneration by transplanting human islets from young and aged donors into immunodeficient mouse models. In addition, it will be interesting to further investigate the effect of combining a GPR119 agonist with a DPPIV inhibitor on reversing autoimmune diabetes.
study demonstrating the effects of a GPR119 agonist along with a DPP-IV inhibitor on b-cell regeneration via both replication and neogenesis in a diabetic mouse model. Besides b-cell regeneration, other factors including prevention of b-cell apoptosis may have also contributed to the increase of b-cell mass. Pancreatic b-cell mass was remarkably improved by combination therapy, which offers a novel therapeutic strategy for treating diabetic patients with a low b-cell mass. As there are clear discrepancies between rodent and human b-cell regeneration capacity, our future studies are aimed at evaluating the potential of these drug combinations on human islet regeneration by transplanting human islets from young and aged donors into immunodeficient mouse models. In addition, it will be interesting to further investigate the effect of combining a GPR119 agonist with a DPPIV inhibitor on reversing autoimmune diabetes.
Consistent with our finding that only one double strand break is formed in minichromosome DNA in irradiated cells
This assumption is not supported strongly by experimental evidence and does not take into account the variable conformations and microenvironments of chromatin in the nucleus. Single or double BIBW2992 strand breakage of minichromosome DNA by apoptotic or other endogenous nucleases did not appear to be significant during incubation of cells for repair. Supercoiled DNA in non-irradiated cells showed no significant decrease in its level between 0 h and 2 h. In irradiated cells its level remained identical to that in control cells when topoisomerases or PARP were inhibited, but its stability in the presence of putative repair inhibitors could not be measured since they influenced its reformation by repair pathways. The level of linear minichromosome DNA in irradiated cells remained constant when NHEJ was inhibited, with a TWS119 p-value for the difference in level between 0 h and 2 h of 0.55 for wortmannin and 0.88 for NU7441. To inhibit enzymes involved in repair of strand breaks, we used chemical reagents whose specificity has been well established because in most cases siRNA methodology did not provide sufficient depletion of enzymes. In other studies depletion of PARP-1, DNA ligases, and topoisomerase II was also less than complete and in some cases lethal. Inhibitors of PARP-1 showed no effect on the repair of strand breaks in minichromosome DNA. The precise step in which PARP-1 intervenes in repair remains elusive; the current view is that it is not indispensable for repair of single strand breaks in genomic DNA and its role appears to be indirect, for example by binding to breaks and protecting them from further degradation. In another study using our experimental system published while this manuscript was in preparation, knockdown of PARP-1 did not significantly affect repair of single or double strand breaks. A possible role for topoisomerases I or II in DNA repair has been examined in several studies, but in some cases noncatalytic topoisomerase inhibitors were employed which themselves create strand breaks when DNA is deproteinised and therefore cannot provide evidence for a role of topoisomerases in repair. Topological considerations predict that if nucleosomes do not dissociate completely in the neighbourhood of a strand break, the negative superhelicity which results from DNA wrapping on their surface  would be conserved in the nicked circular and linear forms. Thus after the repair of all breaks, the religated circular form would recover the negative superhelicity of the original circular minichromosome DNA. Our finding that the conversion of linear to supercoiled minichromosome DNA continues at the normal rate when topoisomerases I and II are inhibited by catalytic inhibitors is consistent with this scenario. It appeared paradoxical at first view that repair of double strand breaks in the minichromosome was arrested completely by inhibition of NHEJ, while 20�C30% of the breaks appeared to be repaired by HR as deduced from the effects of inhibiting activation or activity of ATM kinase or depleting Rad51. These findings can be interpreted plausibly by the mechanism which has been proposed to understand similar observations on repair of double strand breaks in genomic DNA, which is reported to be completely inhibited when NHEJ is arrested by the DNA-PKcs inhibitor wortmannin ; trapping of factors involved in NHEJ at DNA extremities is suggested to prevent the access of factors required for HR.
would be conserved in the nicked circular and linear forms. Thus after the repair of all breaks, the religated circular form would recover the negative superhelicity of the original circular minichromosome DNA. Our finding that the conversion of linear to supercoiled minichromosome DNA continues at the normal rate when topoisomerases I and II are inhibited by catalytic inhibitors is consistent with this scenario. It appeared paradoxical at first view that repair of double strand breaks in the minichromosome was arrested completely by inhibition of NHEJ, while 20�C30% of the breaks appeared to be repaired by HR as deduced from the effects of inhibiting activation or activity of ATM kinase or depleting Rad51. These findings can be interpreted plausibly by the mechanism which has been proposed to understand similar observations on repair of double strand breaks in genomic DNA, which is reported to be completely inhibited when NHEJ is arrested by the DNA-PKcs inhibitor wortmannin ; trapping of factors involved in NHEJ at DNA extremities is suggested to prevent the access of factors required for HR.